

Personal Protective Equipment
Healthcare professionals are both barriers to and carriers of infection. To avoid distributing infection among patients, healthcare professionals must remain diligent in the use of personal protective equipment, or PPE. Gloves, gowns, shoe covers, caps, masks, respirators, hair covers, face protection, and eye protection are all vital in preventing the spread of infection. Nurses who use PPE effectively help prevent the spread of infection through the touch of a hand, through contact with their uniform, and through air and droplets from their respiratory tract. In turn, they are personally protected against any organisms the patient may carry.
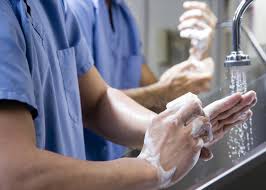
Gloves are the most frequently worn item of PPE. Gloves reduce the opportunity for microbes on healthcare workers’ hands to be spread to patients during care or invasive procedures. Removing gloves between patients prevents the transmission of bacteria from patient to patient. It is important for healthcare professionals to understand that gloves should not be used in place of handwashing. After removing gloves, healthcare professionals should always follow handwashing guidelines.
Gowns and masks also help prevent HAIs. Gowns worn in patient rooms and in operating rooms prevent infectious microbes from being carried from patient to patient on healthcare workers’ clothing. Masks are used to prevent microbes from being transmitted via the respiratory tract. In 2011, the Food and Drug Administration (FDA) approved a new type of N95 respirator for single-use that kills methicillin-resistant Staphlococcus aureus (MRSA), Streptococcus pyogenes, and Haemophillus influenzae. This specialty mask incorporates an antimicrobial agent into the fiber of the mask. The mask eliminates 99.99% of bacteria on its surface within 1 hour and traps and kills additional microbes in the mask’s middle filtration layers.
Transmission-Based Precautions
The topic of PPE is not complete without a review of contact, droplet, and airborne precautions. Most nurses are familiar with the types of precautions used to prevent infection transmission. Even so, nurses, as well as other healthcare professionals, can become lax in their use of precautions, increasing the incidence of HAI.
Contact precautions are designed to reduce the risk of transmission of infectious organisms by direct or indirect contact with the patient or the patient’s environment. Transmission can occur through direct skin-to-skin contact from an infected person or host or through indirect contact of a susceptible host with a contaminated intermediate object or fomite in the environment. To comply with contact precautions, healthcare professionals should follow these guidelines:
- Wear gowns and gloves for all patient contact or contact with potentially contaminated areas of the patient’s environment (e.g., bedrails, furniture, medical equipment).
- Put on PPE before entering the patient’s room and remove it before leaving the room to contain any potential microbes.
- Whenever possible, patients on contact precautions should be admitted to private rooms. Infection control personnel should be consulted before cohorting patients if a private room is not available.
- Transport patients only when necessary for diagnosis or treatment and weigh the risk of infection against the need for transport. When transporting a patient, place the patient in a clean gown and cover the patient with linen per facility policy.
Transmission of microbes by droplet involves contact of conjunctivae of the mucous membranes of the nose or mouth of a susceptible person with large particle droplets. Droplets are generated from the source patient primarily during coughing, sneezing, or talking or during certain procedures, such as suctioning and bronchoscopy. Transmission of infection by large particle droplets requires close contact between the source patient and the susceptible host. Droplets do not remain suspended in the air and generally travel only short distances–up to 3 feet. For this reason, special ventilation systems and air handling are not required for droplet precautions. To properly follow droplet precautions, healthcare professionals should follow these guidelines:
- Wear a surgical mask for close contact (within 3 feet) with patients on droplet precautions; respirators are not necessary.
- Admit the patient to a private room, if possible. Infection control personnel should be consulted before cohorting patients if a private room is not available.
- When transporting patients on droplet precautions, place a mask on them and instruct them in proper cough etiquette.
Airborne precautions are designed to prevent transmission of infectious organisms that remain infectious over long distances if they become suspended in the air. Airborne transmission occurs when there is dissemination of either airborne droplet nuclei (small particle residue of evaporated droplets) or dust particles containing the infectious agent. Airborne organisms are transmitted by air currents and may be inhaled or deposited on a susceptible host within the same room as a source patient or even a long distance away from the source patient. To prevent airborne transmission, healthcare professionals should follow proper airborne precautions:
- Admit the patient to a room with special air handling and ventilation systems. These airborne infection isolation rooms (AIIRs) use negative airflow relative to hallways or surrounding areas to prevent air from escaping the room. The rooms complete 12 air exchanges per hour in buildings with new construction and renovation or 6 air exchanges per hour in existing facilities. Air exhausted from AIIRs must be directly exhausted to the outside or put through a HEPA filtration system before returning to the building.
- Don the proper PPE when entering an isolation room, including an N95 mask–a mask approved to filter 95% of airborne particles. Healthcare professionals must not confuse N95 respirators with surgical masks. They are not the same. Surgical masks cannot filter small particles and do not prevent leakage around the edges when the user inhales. To identify an N95 mask, look for the manufacturer’s name, part number (P/N), the protection provided by the filter, and the letters NIOSH or the NIOSH logo written on either the outside front, the exhalation valve, or the straps of the mask.
- Be sure to be fit-tested for an N95 mask before you care for anyone requiring airborne precautions. Facilities that have AIIRS must have a facility-wide respiratory protection program that includes education of workers on respirator use with appropriate fit testing and user seal checks.
- Limit patient transport to medically necessary procedures only. If the patient must be transported, skin lesions must be covered and the patient must wear a surgical mask and observe respiratory hygiene/cough etiquette.
Environmental Decontamination
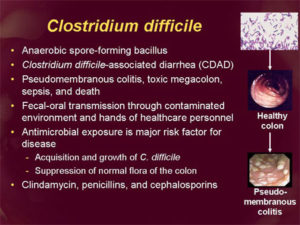
Some bacteria do not need a living host to survive. Microbes such as MRSA, vancomycin-resistant enterococci (VRE), and C. difficile can survive for long periods on environmental surfaces, such as bedrails and phones. After being contaminated, these environmental surfaces become the source of infection. A clean healthcare environment is essential for prevention of infection from these organisms.
Cleaning and disinfection of the patient environment includes high touch surfaces, such as bedrails, carts, toilets, and doorknobs, as well as general housekeeping surfaces, such as floors, walls, and blinds. Proper disinfection and sterilization of medical and surgical instruments and devices are also vital in the prevention of HAIs.
Agents used by facilities that are designated by the Environmental Protection Agency (EPA) as hospital-grade detergents/disinfectants must be able to inactivate specific organisms, such as staphylococci and streptococci. These agents must be chosen carefully to determine if they are effective in killing pathogens.
Nurse Staffing and Burnout and HAI
A recent study in the American Journal of Infection Control showed a correlation between nurse staffing ratios, nurse burnout, and HAIs. According to the researchers, the higher the nurse-patient ratio, the higher the likelihood of occurrence of a HAI due to increased demands on nurses and resulting burnout. The study showed that increasing a nurse’s patient load by one patient increased the incidence of urinary tract infections and SSIs. The authors of this study suggest that healthcare facilities decrease nurse burnout to decrease the incidence of HAIs.
Surveillance of HAIs
Infection surveillance helps experts identify infectious disease, spot trends in infections, and create treatment and eradication goals. The federal government provides resources to estimate and track HAIs affecting patients and healthcare personnel. The National Healthcare Safety Network (NHSN), the largest HAI reporting system in the United States collects data from participating institutions, including acute care hospitals, long-term acute care hospitals, psychiatric hospitals, rehabilitation hospitals, outpatient dialysis centers, ambulatory surgery centers, and long-term care facilities. Currently, more than 9,000 institutions participate in the NHSN to share infectious disease data in a timely manner. As the scope of HAI reporting is extended, it is the goal of the NHSN to target prevention, improve patient outcomes, and reduce healthcare costs, with the ultimate goal being elimination of all HAIs.
Another national resource, the Emerging Infections Programs (EIP), consists of a network of state health departments and their academic medical center partners. These agencies collaborate to answer questions regarding emerging HAI threats, advanced infection-tracking methods, and antibiotic resistance in the United States.
Conclusion
The medical profession has come a long way in its progress toward elimination of HAIs, but there is still a long way to go. Understanding common pathogens, knowing risk factors of HAIs, and implementing preventative measures are key ways healthcare staff can help keep patients safe. Diligence and attention to proper hand hygiene, environmental disinfection, use of proper transmission-based precautions, and the correct use of PPE will help prevent the spread of these infections. With perseverance, healthcare workers can be instrumental in reaching the ultimate goal of zero HAIs.
Resources for Part I throught V.
For more information on isolation precautions including standard and transmission-based precautions, refer to the NCCE course titled Science of Infection Control Principles available at www.nursece.com.
For general information on common HAIs, visit the CDC’s website at www.cdc.gov.
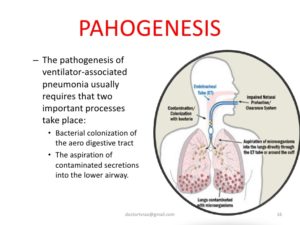
For more information on prevention strategies for VAP, refer to the publication Strategies to Prevent Ventilator-Associated Pneumonia in Acute Care Hospitals, available at http://www.jstor.org/stable/10.1086/591062.
For more information on isolation precautions, see Guidelines for Isolation Precautions: Preventing Transmission of Infectious Agents in Healthcare Settings 2007, available at www.cdc.gov.
For more information on infection control in the LTCF, see the SHEA/APIC Guideline: Infection Control in the Long-Term Care Facility, available at http://www.jstor.org/stable/10.1086/592416.
For more information on developing strategies for measurement of hand hygiene, refer to the Joint Commission’s monograph titled Measuring Hand Hygiene Adherence: Overcoming the Challenges, available at www.jointcomission.org.
For more information on the selection and proper use of disinfection and sterilization materials, see the Guideline for Disinfection and Sterilization in Healthcare Facilities, 2008, available on the CDC website at www.cdc.gov.
Top recommendations and detailed tool kits for preventing healthcare–associated infections, including urinary tract, surgical-site, Clostridium difficile, and central line–associated bloodstream infections outside the ICU are available at http://www.cdc.gov/hicpac/pdf/guidelines/bsi-guidelines-2011.pdf.