 human lungs
human lungs
The respiratory system consists of the lungs, throat, diaphragm, nose and mouth. Working together, these organs convert the air that is breathed into oxygen for the blood.
The process begins when a person breathes in air. Once taken into the body, the air travels through the throat into the chest cavity where it is processed by the bronchial tubes into the alveoli of the lungs. There, unneeded air such as carbon dioxide is removed and prepared to exit the body.
If one of these components of the respiratory system fails, oxygen intake is compromised, and the result can be serious if not fatal.
The major organ used in respirations is our lungs. Your lungs are organs in your chest that allow your body to take in oxygen from the air. They also help remove carbon dioxide (a waste gas that can be toxic) from your body. The lungs‘ intake of oxygen and removal of carbon dioxide is called gas exchange. Gas exchange is part of breathing.
To make this more simplistic in understanding how vital this organ is let us look at the car. It operates mainly with the engine but also works with a transmission without either the car would fail in operation, including the oil system. Let’s look at the body; remember both the car and the human body operate on systems, not one unit. Our body’s engine is the heart with our transmission is the lungs; one completely without the other would die in about less 7 minutes. The renal system (kidneys) is our oil system that filters our blood taking toxics out via the kidneys dumping into our urinary bladder to void out our system (the toxics are the yellow in our urine). Get the idea of how our body is similar to the car.
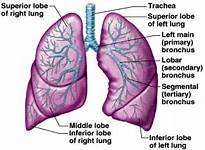
Without losing our topic here lets now continue with anatomy of lungs. The lungs are pyramid-shaped, paired organs that are connected to the trachea by the right and left bronchi; on the inferior surface, the lungs are bordered by the diaphragm. The diaphragm is the flat, dome-shaped muscle located at the base of the lungs and thoracic cavity. The lungs are enclosed by the pleurae, which are attached to the mediastinum. The right lung is shorter and wider than the left lung, and the left lung occupies a smaller volume than the right. The cardiac notch is an indentation on the surface of the left lung, and it allows space for the heart. The right lung has 3 lobes whereas the left has 2. How does this deal with pneumonia well let us know get into this diagnosis.
What is pneumonia?
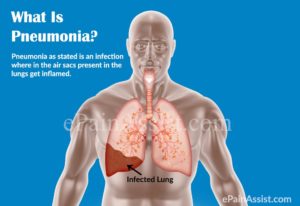
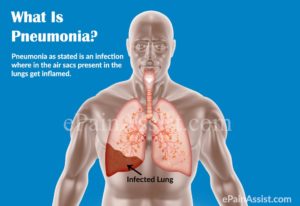
Pneumonia is a lung infection that can make you very sick. You may cough, run a fever, and have a hard time breathing. For most people, pneumonia can be treated at home. It often clears up in 2 to 3 weeks. But older adults, babies, and people with other diseases can become very ill. They may need to be in the hospital.
You can get pneumonia in your daily life, such as at school or work. This is called community-associated pneumonia. You can also get it when you are in a hospital or nursing home. This is called healthcare-associated pneumonia. It may be more severe because you already are ill. This topic focuses on pneumonia you get in your daily life.
What causes pneumonia?
Germs called bacteria or viruses usually cause pneumonia.
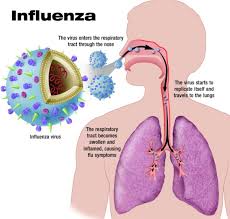
Pneumonia usually starts when you breathe the germs into your lungs. You may be more likely to get the disease after having a cold or the flu. These illnesses make it hard for your lungs to fight infection, so it is easier to get pneumonia. Having a long-term, or chronic, disease like asthma, heart disease, cancer, or diabetes also makes you more likely to get pneumonia.
What are the symptoms?
Symptoms of pneumonia caused by bacteria usually come on quickly. They may include:
- Cough. You will likely cough up mucus (sputum) from your lungs. Mucus may be rusty or green or tinged with blood.
- Fever.
- Fast breathing and feeling short of breath.
- Shaking and “teeth-chattering” chills.
- Chest pain that often feels worse when you cough or breathe in.
- Fast heartbeat.
- Feeling very tired or very weak.
- Nausea and vomiting.
- Diarrhea.
When you have mild symptoms, your doctor may call this “walking pneumonia.”
Older adults may have different, fewer, or milder symptoms. They may not have a fever. Or they may have a cough but not bring up mucus. The main sign of pneumonia in older adults may be a change in how well they think. Confusion or delirium is common. Or, if they already have a lung disease, that disease may get worse.
Symptoms caused by viruses are the same as those caused by bacteria. But they may come on slowly and often are not as obvious or as bad.
How is it treated?
If pneumonia is caused by bacteria, your doctor will give you antibiotics. These almost always cure pneumonia caused by bacteria. Be sure to take the antibiotics exactly as instructed. Do not stop taking them just because you feel better. You need to take the full course of antibiotics.
Pneumonia can make you feel very sick. But after you take antibiotics, you should start to feel much better. Call your doctor if you do not start to feel better after 2 to 3 days of antibiotics. Call your doctor right away if you feel worse.
There are things you can do to feel better during your treatment. Get plenty of rest and sleep, and drink lots of liquids. Do not smoke. If your cough keeps you awake at night, talk to your doctor about using cough medicine.
You may need to go to the hospital if you have bad symptoms, a weak immune system, or another serious illness.
Pneumonia caused by a virus usually is not treated with antibiotics. Sometimes, antibiotics may be used to prevent complications. But home treatment, such as rest and taking care of your cough, usually is all that is done.
How can you prevent pneumonia?
Experts recommend immunization for children and adults. Children get the pneumococcal vaccine as part of their routine shots. Two different types of pneumococcal vaccines are recommended for people ages 65 and older. If you smoke, or you have a long-term health problem, it’s a good idea to get a pneumococcal vaccine. It may not keep you from getting pneumonia. But if you do get pneumonia, you probably won’t be as sick. You can also get an influenza vaccine to prevent the flu, because sometimes people get pneumonia after having the flu.
You can also lower your chances of getting pneumonia by staying away from people who have a cold, measles, or chickenpox. You may get pneumonia after you have one of these illnesses. Wash your hands often. This helps prevent the spread of viruses and bacteria that may cause pneumonia.
You are more likely to get pneumonia if you:
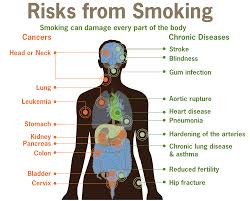
- Smoke. Cigarette smoking is the strongest risk factor for pneumonia in healthy young people.
- Have another medical condition, especially lung diseases such as chronic obstructive pulmonary disease (COPD) or asthma.
- Are younger than 1 year of age or older than 65.
- Have an impaired immune system.
- Take medicine called a proton pump inhibitor (such as Prilosec or Protonix) that reduces the amount of stomach acid.
- Drink excessive amounts of alcohol.
- Recently had a cold or the flu.

 human lungs
human lungs
